How to freeze to death in Wyoming in the summertime
and other paradoxes of radiation
A seminar presented March 14, 2001
at LCCC Star Club, Cheyenne, Wyoming
by
Dr. Kevin T. Kilty
Those of you who have taken a two semester course in general physics or even a
two semester course in engineering physics may have gotten some introduction to the
topic of this seminar. You know that all objects warmer than absolute zero radiate
electromagnetic radiation. You were probably introduced to the Stefan-Boltzman law, which relates
the total power radiated from a hot object, per unit area of surface, and into a
2p steradian solid angle, to the temperature of the object. For
those of you not familiar with solid angles, a solid angle
of 2p is a hemisphere. In other words, this represents the entire
view above a plane surface; or, if you wish, this is the biggest view in which
none of the plane can "see" any portion of itself. Stefan's Law is
Power=seT4; where T is the absolute temperature
of the body and two leading constants represent, respectively, the Stefan constant
and the emissivity of the body; e=1 applies to a special type of surface known as a blackbody.
In some instances you may have seen Stefan's Law modified a little bit to account for
radiation coming toward the object from its surroundings. Letting To stand
for the "effective" black body temperature of the surroundings, the modified law is
Power=se(T4-To4)
This at least acknowledges the importance of the surroundings, but is otherwise pretty
worthless because we have no means for calculating To!
Perhaps you also learned of Wein's displacement law, which states that the peak
wavelength of this broadband thermal emission is proportional to 1/T. In fact,
if we measure wavelength in units of micrometers, then a good approximation is
l=3000/T. Thus, the sun at 6000K has a peak
wavelength just about where our eyes detect green light at
0.5m, a human being at 300K radiates with peak power near
10m, and the cosmic background at 3K has a peak in its
spectrum near 1mm.
More than likely this is all you learned. What you didn't learn is that what you did
learn is completely inadequate to work the simplest of heat transfer problems
involving radiation.
What an engineer would like to know.
Engineers have to solve practical problems. Their solutions are tested when a
design is first put to use. This means that they can't solve the sort of wimpy
problem we typically find in a physics textbook--something like "what is the heat
radiated from a one-liter teapot filled with boiling water?" But armed only with Stefan's Law, this sort
of problem is the extent of what a person can solve. Engineers need to find answers
to the following types of questions:
- How much heat will an object transfer through radiation to some other object?
- What will be the equilibrium temperatures of a collection of objects in a field of radiation?
- How will the temperature of some object evolve?
- What will happen if I alter a design?
How do we go about answering such questions?
In order to answer any such questions we have to modify and extend the Stefan Law because it deals only with the
emissive power of a hot object. We can do this as follows.
- First, we ask, "How
will objects absorb and reflect radiation?" From the first law of thermodynamics,
or conservation of energy, if you will, it is apparent that the total of reflected,
absorbed, and transmitted energy must be constant. There is no other place for energy to go.
Therefore, using r, a, and t
as the coefficients involved, the first law is r + a + t = 1.
From the second law of thermodynamics
it is true that a = e, this is known as Kirchoff's
Law. You can verify this by noting that you cannot distinguish objects, no matter
how different they may be from one another, if they are isothermal--inside of something
like a kiln, for example.
Strictly speaking all of these coefficients vary with wavelength, so Kirchoff's Law
should actually read...
al = el
- Next, to account for the disposition of all energy transferred among or between
objects we need to define...
- J=Radiosity which is the total radiant power leaving the surface of an object.
Not all of this has the object emitted. Some is reflected from other objects or transmitted from behind.
- G=irradiation which is the total radiant energy falling on a surface.
- W=emissive Power which is the total power emitted by the object by way
of its absolute temperature.
Of course, it goes without saying that all of these quantities are per unit area of surface
and per hemispherical solid angle.
An example relationship among these is J=W+rG, which holds
for objects that are not transparent to radiation. I have an example showing the distinction
between radiosity and emissive power.
- Third, because all quantities are stated per unit surface area
and per solid angle of the view, then heat transfer depends on both surface area
and orientation of surfaces with respect
to one another. In other words, there are ugly four-dimensional integrals lurking about.
For instance, figure 1 shows a series of surfaces where (1) is a radiator and (2)
is an absorber. Despite the surface areas involved being the same in each case, the
heat transfered q1->2 is different for each because the view of one another
is different. Engineers generally hate to do four-dimensional integrals, so
the more mathematically inclined among them will always compile tables of factors for
various shapes and orientations. Most engineering texts on heat transfer contain
such tables.

- Finally, to account for behavior of materials which are not ideally black, we
allow that el<1 and that it varies according to
material. The actual values have to be found from experiments.
For a blackbody we can use these methods to modify Stefan's equation to pertain
to two surfaces which are exchanging radiation. Let q1->2 be the total
heat transfered from (1) to (2) per unit time. Let F1->2 be a heat transfer factor, which
engineers call a shape factor. This is that four-dimensional integral we can read from a table. Let T1
and T2 be the absolute temperatures of the two surfaces. Then
q1->2=A1F1->2s(T14 - T24)
This equation is far more useful than even the modified form of Stefan's Law because
it tells us the heat transferred between two specific surfaces at specific temperatures.
In other words, between two objects in a design.
If the objects in question are not black, but grey, then rather than use
absolute temperature we use radiosity. The radiant exchange from surface
(s) to (t) is
qs->t=AsFs->t(Js- Jt)
If there are more surfaces involved than two, then the above equation simply contains more terms
on its right hand side, and there are equations for each object. In this case the solution involves a system of
equations.
Note the following very useful fact. If we think of q as a "current," J as a "voltage," and 1/(AF) as
a "resistance," then the problem of finding the equilibrium heat transfer rates
among a collection of objects is just like a DC electrical network problem. If we seek
equilibrium radiosities (or temperatures), instead, the problem is an inverse
DC electrical network problem. In other words, any sophomore engineering student
who had taken my intro to engineering 1610 course, could work such problems.
In particular, they could explain the paradoxes which follow.
Some paradoxes involving radiation.
The firetruck fiasco.

In the autumn of 1979 a condominium building, which was under construction, caught
fire in north Cheyenne. When the firemen arrived at the scene, there was one end of the
building which was not on fire, and they proceeded to pull their firetruck -- a nice, new,
very expensive firetruck -- alongside of this wall.
I do not know why what happened next did happen. I'm sure there was some inquest at a later time, but
I do not know its findings. What happened was that the entire building, including the wall
adjacent the firetruck, being a tinderbox, was soon an inferno. This is entirely expected in an open
wooden structure. What I do not know is why the firemen failed to notice that the entire building was about
to be engulfed in flames, and why they failed to take their truck a safe distance away. Perhaps, and
this is entirely understandable, they reasoned as follows. Their experience is that it is safe to stand twenty
feet from a roaring fireplace, and since this inferno is no hotter than such a roaring fireplace, then twenty
feet is a safe distance for our truck. With nothing more than Stefan's law as
guidance one could easily misapprehend the implication of solid angle in the formula.
In brief, a roaring fire at twenty feet subtends a very small solid angle; a blazing building
at twenty feet is something different altogether.
To gain an appreciation for this situation, let's perform an approximate calculation
of the radiative transfer from the building to the fire truck. Let's assume that the
wall was 40 by 20 feet in size, as in Figure 2, and also a blackbody. I don't know if this is true, but it can't be far wrong. The
wall at this time was a mass of bright orange/red gases, soot, and glowing, charred
wood. If the emissivity of such a thing is not 1.0, it isn't likely to much much lower. Likewise, assume
the firetruck is roughly 8 by 20 feet in size, and also a blackbody. I know that it is actually red, and has plastic, chrome,
and brass parts along its side, but such materials become quite hot in the midday
sun, and we may as well consider such stuff black. These two black panels faced one another
at a mere 20 foot distance. You may go through the tedious, but not difficult calculations
yourself, but the geometry of this situation, as read from a table, provides a form factor of approximately 6.00
square meters. If we consider that the firetruck was initially at 300K and the fully
developed blaze somewhere between 1000K and 1250K, the transfer rate from the fire
to the truck was somewhere between 320,000 and 660,000 watts.
We now appreciate the predicament of the firemen. Their pumper was nearly out
of water, and the driver's side of the truck was searing in radiation equal to 30 or maybe 60 summertime midday suns. It got hot very fast. The plastic melted.
The paint bubbled and peeled. The tires caught fire and exploded. The truck sat next to the burnt
out ruin of the condos for a week or more as a sort of monument to misunderstanding solid angle.
What's the heat-flow deal if we are inside? Why do heated rooms feel cold?
Everyone knows that there is something "funny" about building temperature control.
A thermostat in any building is placed, by statute, where it can't measure
the effective temperature of the room. Figure 3 shows a room with a very large window, providing
a wonderful view. The outside air temperature is 0°C (273K), the inside surface of the window
is 10°C (283K--not at all unrealistic) and the air temperature, and all other surfaces, in the
room are 26°C (299K), just what the thermostat orders. There is a hard-working executive, practicing his putting,
at the window. What temperature does this fellow feel?
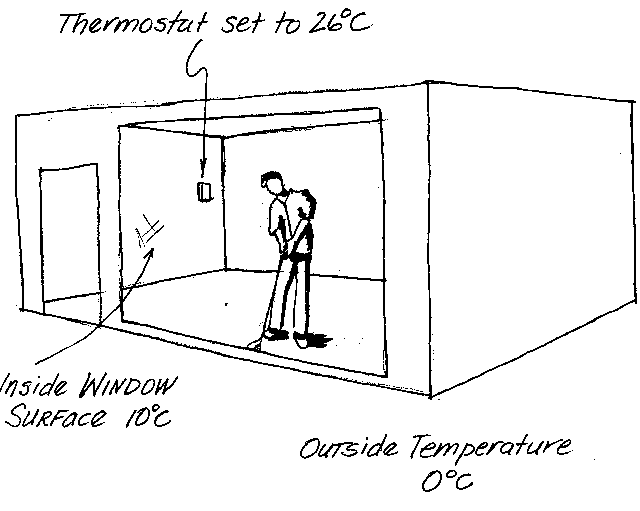
It all depends on what the window is like. If it is completely transparent to infrared
radiation (e is nearly zero) then the window and the person
will transfer heat between them like the window is 0°C (273K). Since the window is very large,
it forms nearly a hemispherical view. Meanwhile, the remainder of the room is a hemispherical
view of a blackbody at 26°C (299K). On average the person transfers heat to the room
like it is a mere 13°C (286K). The room may even feel colder because people are quite sensitive
to small surface areas of skin that are too cold.
If, on the other hand, the window is made of one of those high e
glasses like PPG's Sungate®, which are coated with stannous oxide, then it will
radiate near its actual temperature (283K). Yet the room may still feel too cold. In this case,
what room comfort probably requires is a double paned glass, which allows the inside surface to
reach a temperature near the thermostat setting. In fact, in this case we may be able to lower
the thermostat because the room will have no cold spots. The important point to remember is that air temperature can be a poor
guide to comfort.
What's the heat transfer thing if we are outside?
The natives in many dry climates can make ice at night, even when the air temperature
remains near 55°F, by exposing a shallow earthen dish of water on a high piece of ground
to a clear, dry, night-time sky. In July 1995, my brother found a dead person lying atop a hill. He,
too, had been exposed to a clear, dry, night-time sky; and the sheriff concluded that the person
had died of exposure on a night when the air temperature in all likelihood stayed near 50°F all night.
What do these have in common?
Figure 4, in the upper part, shows a person outside in sunshine. It is well known that
about 40-50% of heat loss from a human occurs through radiation. Therefore,
radiation is very important for maintaining our body temperature. During the day
there is a 6000K sphere in view that is 0.5°of arc. It is the sun. There is diffuse radiation
coming from other parts of the sky as well. Also, the earth,
and vegetation are warmed by the sun, and all radiate also. Thus, even if the air is
not particularly warm, a person can feel comfortable on a sunny day because some object at every
possible view is radiating.

Now let us examine the same situation at night. The person, having a nearly constant
surface temperature radiates at the same rate as before. Skin, by the way, is nearly
a blackbody at long wavelengths(e=1). But now there is no
sun to radiate energy, there is little or no diffuse light from the sky, and the earth
and plants are all much cooler. Does any radiation come from the sky?
The answer depends on what is in the sky. Probably the most important thing is
how much water vapor is in the air? Water vapor is a very important absorber of
infrared radiation, which means also that it is a very important emitter. If the air near
the ground surface is very wet, then a thick layer of air will radiate just like a blackbody
of the same temperature. This is why the evening cools-off so little in places like the
Southeast or Midwest. If, on the other hand, the air is quite dry, as it is in Wyoming or in desert areas,
then it absorbs and radiates almost nothing. In this case that 50% radiated energy from the body is a
very substantial heat loss, which has to be is replaced from within the body. Is the body
up to the task?
The dead person my brother found on the hilltop, had gotten his car stuck in mud
during the night. As he struggled to free the car he apparently had beer to drink--quite a lot
of beer i think--and he also became very warm. As he continued to wrestle the car he took
off clothing. Finally he wandered up a nearby hill, with substantial blood alcohol, nearly naked, and passed out on the hill top.
Put as simply as possible the chain of causation here is...
Wyoming-->Dry, Clear, Night-time sky-->Go naked-->Die.
Keep your clothes on, folks, they make a good radiation baffle!
The katabatic wind: or heating by cooling.
This is not a paradox of radiation per se, but it involves radiation along the same lines
as the previous paradox. Consider how air, near the ground surface,
behaves on a summer night in Wyoming. The ground surface radiates to the night sky, cools rapidly,
and cools the neighboring air. We have just seen that this occurs in a most pronounced way when the
air is dry, as it often is in Wyoming. As this air cools it does so near a particular ground elevation.
In other words, it cools at constant pressure. We can organize the ideal gas law in
this situation to...
rT=constant.
The air becomes more dense as it cools along an isobar. In figure 5 as
the air cools it
follows the iosbar (a hyperbola) from 1 to 2. The ground probably slopes in some direction,
and the air, having now become more dense flows downslope to lower elevation. It is
extremely difficult to maintain static air on sloping ground when there is radiation
heat transfer, and this is one reason (among millions of others) why Wyoming is windy. This
wind is called a katabatic wind.
In order to understand what happens as air sinks downhill, you first must
recognize that air pressure increases with lowering elevation. The reason, simply put, is
that air pressure has to support the weight of all of the air above, and at lower elevation there
is more air mass to support.
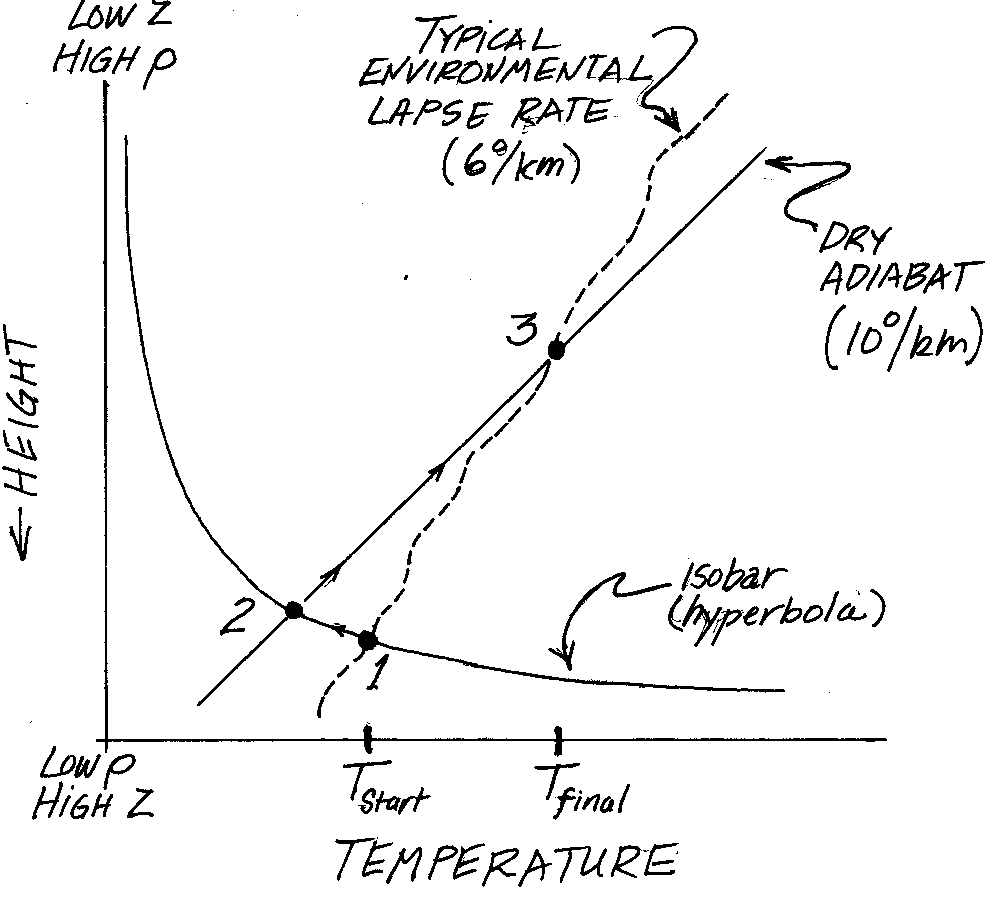
What happens to the parcel of air that became too dense and is now sinking down a
sloping surface? Well, its pressure certainly rises, but what else occurs? The answer
could become very complicated, but if we assume it sinks
so rapidly that it cannot transfer heat, then the first law of thermodynamics
tells us what occurs. The first law is dU=dq-dw.
I know you didn't fully understand this at the time, but dU is an exact differential
which is a difference of two inexact ones. The inexact differentials we can calculate only
when we know how heat is transfered or work is done. In this particular case we set dq
equal to zero because we are not transfering heat. The inexact differential
dw is quite easy to state--if we use heat content (H)
rather than internal energy (U), then the only work involved is mgdz; where
m is a mass of air (who cares how much?) and g is gravitational acceleration. Ah,
you will recognize no doubt, that work in this case is just like lowering a mass in
physics lab. Meanwhile, the exact differential we can calculate independently of how
the air arrived at its current state. Being an ideal gas (ideally that is) its heat
content is just dH=mcpdT. Now reorganize this to dT/dz=-g/cp.
Meteorologists call this the dry adiabatic lapse rate, and it describes how the temperature
of air will change if it is moved quickly to a new elevation. It is the straight line on Figure 5.
The sinking parcel of air follows this line from 2 to 3 on the diagram.
So, we conclude that the sinking air will undergo a change of temperature and a
change of pressure, which means its density changes. Moreover,
it will not stop flowing downhill until its density equals that of the surrounding air.
When will this occur, you ask? Figure 5 also shows a sort of ragged line that is meant to
indicate how the state of the surrounding air varies with height. Meteorlogists call
it the environmental lapse rate, and it has a value near 6C/km.
You might ask, why does the environmental lapse rate not equal the adiabatic rate?
This is a good question, because it is easy for me to answer. The reason is that the envirnoment
has evolved to its current state though a whole series of unknown processes, or,
in other words though a whole bunch of steps of dq and
dw, and the environmental lapse rate simply represents the end
result of it all.
On Figure 5 I show that the sinking air stops sinking where
these two curves cross. Always that happens when the sinking air reaches a greater density
and higher temperature than it had when it began radiating early in the evening. The air has
warmed by loosing heat! Isn't radiation weird?
Almost any summer night, you can go to the Goshen Hole country in eastern Wyoming, and feel a
katabatic wind flowing down the drainages of Horse Creek and Bear Creek. In drought
years, when the air is especially dry, this wind is at its most severe, as you'd expect.
In these years the wind can reach 70mph and adds additional misery for crops and
farmers beyond what the drought does already.
A bunch of objects in equilibrium with radiation don't have the same temperature!
You learned in physics that objects at thermal equilibrium all have the same temperature. Yet
if you examine objects bathed in the radiation field of a summer day, objects which
are free to exchange energy and which you'd think of as reaching equilibrium, they aren't the
same temperature at all. After all, the white plastic deck chair can feel cold to your
touch while the red hood of the sports car is hot enough to fry an egg. What trick
is radiation playing now?
Earlier I said that the coefficients describing emissivity, absorbtion, and transmission vary with
wavelength. This makes it possible that one material is very absorptive at a particular wavelength
and not emissive at another; and a different material is not absorptive at the first wavelength, but
very emissive at the second. These two materials will not behave the same.
If we are interested in how materials will behave when in sunlight, then the important figure
of performance is the ratio of absorption coefficient in visible light, as
to emissivity at thermal infrared wavelengths, eb. Examples of
two extreme materials in this regard are aluminum painted with white epoxy paint,
for which the ratio is 0.3, and brushed stainless steel, for which the ratio is 4.3. If
these two materials are placed in full sunlight, and prevented from cooling by any means
other than long-wave radiation, they will achieve equilibrium temperatures of 286K
and 564K, respectively! A Carnot heat engine operating between these two temperatures
would be 50% efficient, far better than any photovoltaic solar panels.
Conclusions
We are out of time, especially so as I planned some time for questions and answers. In summary,
it should be obvious that the subject of radiation heat transfer is very interesting. It
is also very complex, and becomes even more so when absorbing gases are present, and when
solid surfaces transmit radiation and so forth. These few examples, I hope have shown
you how to begin solving realistic, and interesting problems.




